IV Cannula
MOQ : 1000 Pieces
IV Cannula Specification
- Application
- Clinical Use, Hospital Use
- Disposable Or Reusable
- Yes
- Material
- Medical grade PTFE
- Instrument
- Basic Surgical Instruments
- Condition
- New
- Sterilized
- Yes
- Usage
- Infusion Therapy
- Size
- 14 FG to 24 FG
IV Cannula Trade Information
- Minimum Order Quantity
- 1000 Pieces
- FOB Port
- MUMBAI
- Payment Terms
- Paypal, Western Union, Telegraphic Transfer (T/T), Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 5000 Pieces Per Week
- Delivery Time
- 1-2 Week
- Sample Available
- Yes
- Sample Policy
- If order is confirmed we will reimburse the sample cost
- Packaging Details
- Poly Pack, 100/Bax, 1000/Carton
- Main Export Market(s)
- Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Africa, Asia, Central America
- Main Domestic Market
- All India
- Certifications
- ISO,CE
About IV Cannula
Intravenous (IV) cannulation is a technique in which a cannula is placed inside a vein to provide venous access. Venous access allows sampling of blood as well as administration of fluids, medications, parenteral nutrition, chemotherapy, and blood products.
Features:
- Used for intra venous infusion of medicational fluids
- Made from non-toxic Medical grade PTFE
- Formulated cannula tip and smooth surface for atraumatic catheterization
- Super sharp needle with two cutting edges
- Provided with injection port with non-return valve for extra medication
- Application: Infusion Therapy
- Size Available: 14 FG to 24 FG
- Material Used: Medical grade PTFE
- Packing: Poly Pack, 100/Bax, 1000/Carton
Medical-Grade Quality and Safety
Our IV cannula is manufactured using premium medical-grade PTFE and robust plastic, ensuring durability, biocompatibility, and resistance to chemical and physical stresses. Each device is thoroughly sterilized, guaranteeing safety for irrigation and infusion therapies in clinical and hospital environments.
Versatile Sizing for Broad Applications
Available in sizes from 14 FG to 24 FG, our IV cannulas are suited for pediatric, adult, and geriatric patients. This comprehensive range facilitates tailored vascular access for administering fluids, medications, or blood products, making them ideal for daily clinical procedures.
FAQ's of IV Cannula:
Q: How is the IV cannula used in clinical and hospital settings?
A: The IV cannula is inserted into the patient's vein to establish a secure channel for administering fluids, medications, or blood products as part of infusion therapy. Its medical-grade material and sterilized condition make it suitable for routine clinical interventions.Q: What are the main benefits of using a medical-grade PTFE IV cannula?
A: Medical-grade PTFE IV cannulas offer high biocompatibility, reduced risk of allergic responses, and smooth insertion. Their durability and flexibility contribute to a lower incidence of phlebitis or other complications during infusion therapy.Q: When should I select a specific size of IV cannula (14 FG-24 FG)?
A: The choice of cannula size depends on the patient's age, vein condition, and the type of therapy. Larger sizes (14-18 FG) are typically used for rapid fluid or transfusion needs, while smaller sizes (20-24 FG) suit children or patients with delicate veins.Q: Where are your IV cannulas manufactured and distributed?
A: Our IV cannulas are produced in India, adhering to strict quality standards. They are available through leading distributors, manufacturers, suppliers, exporters, traders, and wholesalers, ensuring reliable access for hospitals and clinics nationwide and internationally.Q: What is the process for ensuring the sterilization of each IV cannula?
A: Every IV cannula undergoes rigorous sterilization procedures using advanced technology before packaging. This ensures each unit is free from contaminants and ready for immediate clinical use, supporting patient safety.Q: Can your IV cannulas be reused?
A: Our IV cannulas are designed primarily for single use to minimize the risk of infection. In some controlled clinical scenarios, limited reuse may be allowed in accordance with hospital protocols, but disposable use is strongly recommended.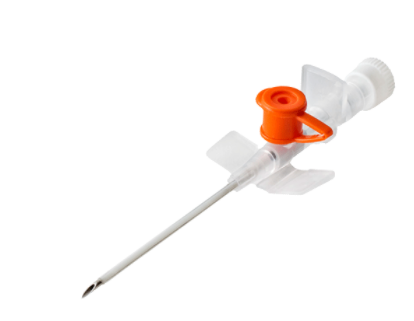
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Surgical Instrument Category
Foley Balloon Catheters
Price 0.50-1 USD ($) / Piece
Minimum Order Quantity : 5000 Pieces
Material : Other, Latex or Silicone
Condition : New
Size : 6 Fr to 26 Fr
Levins Tube
Price 20.0 USD ($) / Unit
Minimum Order Quantity : 100 Units
Material : Other, Medical Grade Polyvinyl Chloride (PVC)
Condition : New
Size : Available in Various Sizes (Adult/Pediatric/French Gauge sizes)
Insulin Pen
Price 10.0 USD ($) / Piece
Minimum Order Quantity : 2000 Pieces
Material : Plastic
Condition : New
Lumbar CSF EDS
Price 20.0 USD ($) / Unit
Minimum Order Quantity : 100 Units
Material : Plastic
Condition : New
Size : 30 cm

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry