Ventricular External Drainage System
MOQ : 1000 Pieces
Ventricular External Drainage System Specification
- Material
- PVC
- Application
- used for managing cerebrospinal fluid (CSF).
- Disposable Or Reusable
- Yes
- Instrument
- Neurosurgery Instruments
- Type
- Ventricular External Drainage System
- Condition
- New
- Sterilized
- Yes
- Use Type
- Wound Drainage After Surgery
- Usage
- Neurosurgery
Ventricular External Drainage System Trade Information
- Minimum Order Quantity
- 1000 Pieces
- FOB Port
- MUMBAI
- Payment Terms
- Telegraphic Transfer (T/T), Western Union, Paypal, Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 5000 Pieces Per Week
- Delivery Time
- 1-2 Week
- Sample Available
- Yes
- Sample Policy
- If order is confirmed we will reimburse the sample cost
- Main Export Market(s)
- Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Africa, Central America, Asia
- Main Domestic Market
- All India
- Certifications
- ISO,CE
About Ventricular External Drainage System
Ventricular External Drainage System contains:
- Bag
- Ventricular Catheter 30 cm
- Stylet
- Connector
- Trocar
- Catheter Anchoring Collar
- Connecting Tube
- Application: Neurosurgery
- Packing: Poly Pack
Precision in CSF Management
This Ventricular External Drainage System provides controlled drainage of cerebrospinal fluid, ensuring the safe and efficient reduction of intracranial pressure after neurosurgical operations. Its advanced mechanism helps clinicians maintain patient safety and monitor health outcomes effectively during critical post-operative periods.
Reliable and Sterile Medical Solution
Made from high-quality PVC and processed for sterilization, the system supports both single-use (disposable) and multiple-use (reusable) applications. Its versatility and compliance with clinical hygiene standards make it suitable for routine neurosurgical procedures in hospitals and other healthcare settings.
FAQ's of Ventricular External Drainage System:
Q: How is the Ventricular External Drainage System used in neurosurgery?
A: The system is inserted into the ventricular cavity to drain excess cerebrospinal fluid (CSF) after surgery. It helps control intracranial pressure, assisting medical teams in managing patients with brain injuries or after neurosurgical interventions.Q: What materials are used in the construction of this drainage system?
A: This system is made from high-grade, medical PVC, which ensures biocompatibility, durability, and resistance to chemical interactions, making it safe for surgical and hospital use.Q: When is it appropriate to use this ventricular drainage system after surgery?
A: It is typically used immediately following neurosurgical procedures where CSF management is essential, such as in cases of hydrocephalus, traumatic brain injury, or post-operative edema.Q: Where can medical facilities acquire this product in India?
A: Hospitals and clinics in India can obtain this system through authorized distributors, exporters, manufacturers, suppliers, traders, and wholesalers who specialize in neurosurgical instruments.Q: What is the process for sterilizing the system?
A: Each system is sterilized prior to packaging, and reusable variants can be sterilized again following standard hospital procedures before reuse, ensuring patient safety and compliance with medical protocols.Q: Is the system available as both disposable and reusable types?
A: Yes, the Ventricular External Drainage System is offered in both disposable and reusable options, providing flexibility for different surgical preferences and institutional policies.Q: What are the primary benefits of using this neurosurgery instrument?
A: The main benefits include effective CSF drainage, reduced risk of infection due to sterilization, adaptability for various clinical needs, and enhanced patient safety during recovery from neurosurgical procedures.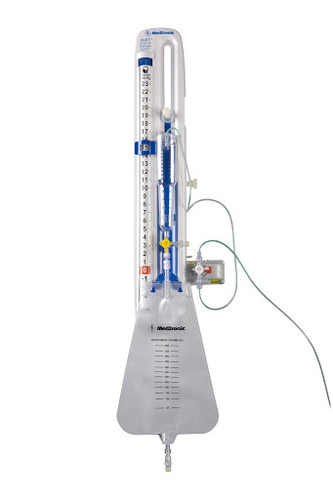
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Surgical Instrument Category
Urine Collection Bags
Price 0.10 INR / Unit
Minimum Order Quantity : 2000 Units
Condition : New
Portable : Yes
Material : Other
Type : Other
Bone Marrow Needles
Price 20.0 USD ($) / Piece
Minimum Order Quantity : 100
Condition : New
Portable : Yes
Material : Other, Stainless Steel
Type : Other, Bone Marrow Needle
Surgical Gloves
Price 1.5 USD ($) / Pair
Minimum Order Quantity : 5000 Pairs
Condition : New
Material : Other, Latex
Type : Other, Surgical Gloves
Aspiration Catheter
Price 250 INR / Piece
Minimum Order Quantity : 1000 Pieces
Condition : New
Portable : Yes
Material : Steel
Type : Other, Aspiration Catheter

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry